This blog post and the “Deep Dive” podcast, created by NotebookLM, are based on “Carbon dioxide overload, detected in human blood, suggests a potentially toxic atmosphere within 50 years” by Larcombe & Bierwirth (2026), and a Physics Today article “Rising carbon dioxide levels now detected in human blood”.
According to a recent study, rising atmospheric CO2 causes human blood pH to decrease, making it more acidic. This process is essentially the human biological equivalent of “ocean acidification.” The drop in pH is driven by a simple chemical reaction that occurs when carbon dioxide enters a liquid environment (like your blood):
- The Reaction: CO2 + H2O → H2CO3 → H+ + HCO3–
- The Mechanism: When you breathe in air with higher levels of CO2, the gas dissolves into the water in your blood to form carbonic acid (H2CO3). This acid then breaks apart, releasing hydrogen ions (H+).
- The Result: In chemistry, the concentration of hydrogen ions determines the pH. The more hydrogen ions present, the lower (more acidic) the pH becomes. This condition is known as respiratory acidosis.
Normally, your body is very good at maintaining a stable pH (around 7.35 to 7.45). When CO2 levels rise, your body uses two main defense mechanisms to stop the blood from becoming too acidic:
- The Bicarbonate Buffer: Your body increases the levels of bicarbonate (HCO3–) to help neutralize the acid. The new study specifically noted that baseline bicarbonate levels in the general population are now trending upward as a compensatory response to the 420+ ppm of CO2 in the atmosphere.
- The Bone “Bank”: To get enough bicarbonate and other minerals to buffer the acid, the body may actually draw calcium and phosphorus from the bones. The study suggests this is why researchers are seeing a steady decrease in blood calcium and phosphorus alongside the rise in CO2.
In the past, these pH shifts usually only happened to people with lung diseases (like Chronic Obstructive Pulmonary Disease) or in poorly ventilated rooms. The significance of the new report is the finding that global atmospheric CO2 has now reached a level where it is creating a “permanent and growing change in human blood chemistry” for everyone, regardless of their health or indoor environment. In summary, our blood is becoming more acidic (lower pH) because the higher concentration of CO2 in the air makes it harder for our lungs to “offload” the gas, causing it to build up in the bloodstream and turn into carbonic acid.
Is the Air Rewriting Our Biology? 5 Surprising Ways Rising CO2 is Already Changing Your Blood
1. Introduction: The Climate Change Inside Us
Stop for a moment and take a deep breath. While we usually view this act as life-sustaining, the very composition of the air you just inhaled is beginning to rewrite your internal chemistry. We have long treated the “greenhouse effect” as an external crisis of melting glaciers and rising tides, but we are overlooking a more intimate transformation: the internal acidification effect.
The human body is no longer a static baseline; it is an active participant in a changing atmosphere. As we pump carbon dioxide into the sky, our blood is mounting a desperate physiological defense. Recent data suggests that the “climate change” occurring within our veins is not a future threat, but a developing reality that is already pushing our biological systems toward their breaking point.
2. Our Blood Chemistry is Trending Toward a “Healthy Limit”
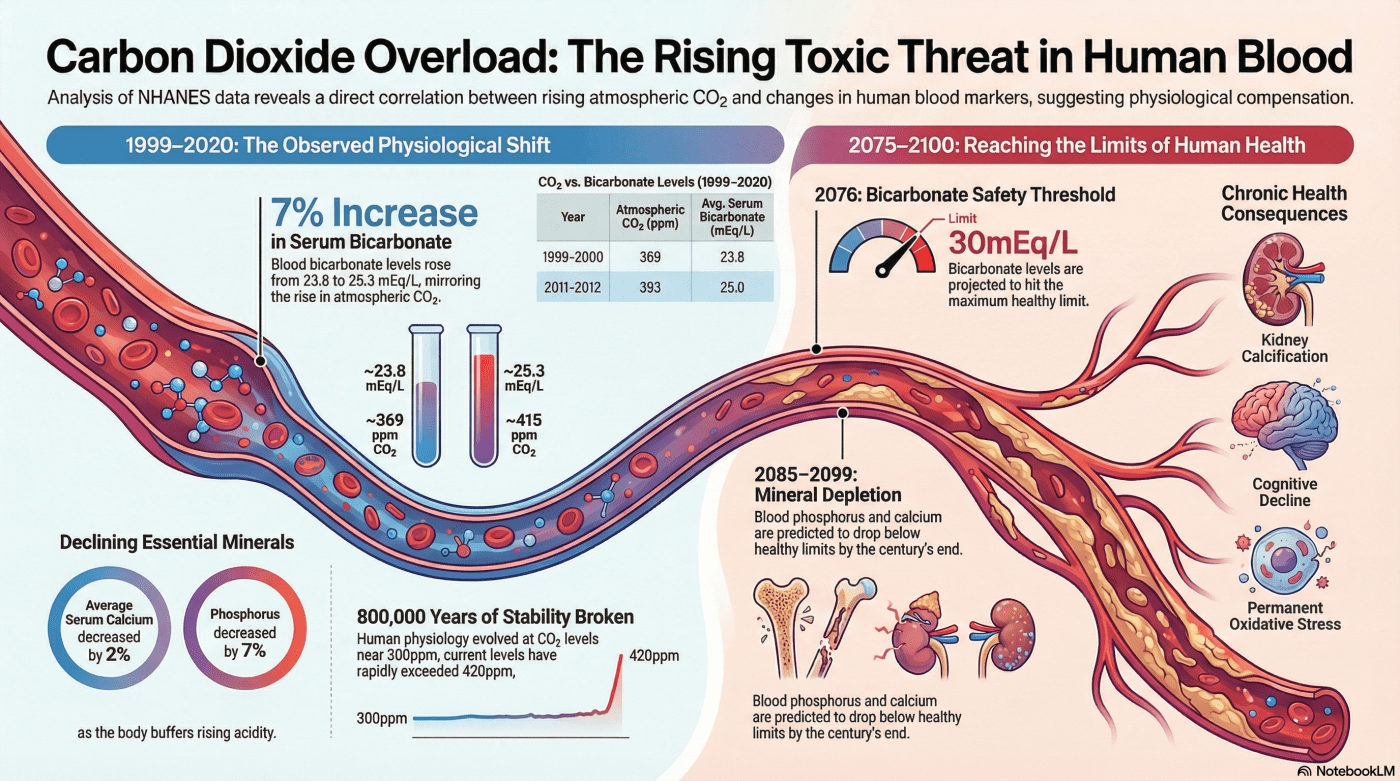
Evidence from the U.S. National Health and Nutrition Examination Survey (NHANES) reveals a startling shift: serum bicarbonate (HCO3−) levels in the American population climbed by roughly 7% between 1999 and 2020. This trend tracks the rise of atmospheric CO2 with haunting precision. Bicarbonate is the body’s primary buffer against acidity, and its surge indicates that our systems are working overtime to neutralize the extra CO2 we inhale with every breath.
We are witnessing a biological irony where the body’s attempt to maintain equilibrium signals a deeper struggle. While medical standards for venous blood consider up to 30mEq/L to be the “healthy maximum,” we are closing the gap at an alarming rate. Based on a linear trendline of a 0.34% annual increase, researchers predict the average person will hit that 30mEq/L limit by the year 2076.
“Assuming a linear relationship… the calculated trendline… predicts that the healthy maximum HCO3− level of 30mEq/L will be reached in the year 2076.”
3. The Quiet Depletion of Essential Minerals
As the body redirects its resources to buffer acidity, other vital markers are plummeting. Between 1999 and 2020, serum calcium (Ca) dropped by approximately 2%, while phosphorus (P) fell by a staggering 7%. This isn’t just a statistical quirk; it is a sign of the body raiding its own stores. To neutralize excess acid, the system pulls minerals from the bone, but chronic exposure eventually leads to hypercalciuria—a state where these vital minerals “leak” out of the body through the urine.
This depletion strikes at the very foundations of human vitality. Phosphorus is a non-negotiable component of ATP (adenosine triphosphate), our cellular fuel, and 2,3-DPG, which is essential for transporting oxygen through the blood. We are effectively trading our bone density and cellular energy levels for the ability to process a more toxic atmosphere.
4. An Evolutionary Mismatch: The Ventilation Blind Spot
For 150,000 years of human evolution, our ancestors breathed air with CO2 levels stable at around 280 to 300 ppm. Our chemoreceptors and ventilation systems were forged in that stability. Today, we have surged past 420 ppm, creating a dangerous evolutionary mismatch. Our biology is “tuned” to an atmosphere that has ceased to exist, leaving us with a critical internal blind spot.
The danger lies in how we breathe. Our lungs do not increase ventilation based on CO2 levels alone; they respond primarily to blood pH. Because our bodies are so efficient at using bicarbonate to buffer that pH, our lungs “think” everything is fine even as CO2 gas accumulates in our tissues. This physiological silence means we aren’t breathing deeply enough to expel the modern overload, leading to permanent CO2 retention.
5. The Mental Cost of Modern Air
The impact of rising CO2 reaches far beyond the blood and into the brain’s delicate circuitry. While outdoor levels rise, indoor environments—where many spend 90% of their time—often trap CO2 at levels between 1,000 and 3,000 ppm. Studies utilizing EEG signals have shown that even at 950 ppm, brain activity heightens and cognitive performance falters.
Beyond simple “brain fog,” CO2 acts as a primary biological alarm. Sensitivity to CO2 is a fundamental survival mechanism; even moderate increases can trigger the autonomic nervous system’s stress response. We are essentially forcing the global population into a low-grade state of biological emergency, which may be subtly fueling modern anxieties and societal conflicts as our nervous systems react to a constant, invisible environmental stressor.
6. The Hidden Danger of “Protein Malfunction”
At the cellular level, the risks become existential. When CO2 levels remain high, the resulting metabolic acidosis can trigger the production of Reactive Oxygen Species (ROS), leading to widespread oxidative stress. This environment causes the calcification of tissues in the kidneys and arteries. Even more concerning is the disruption of the endoplasmic reticulum (ER), the organelle responsible for protein synthesis.
Elevated CO2 and bicarbonate levels trigger “ER stress” and interfere with the charge distribution of molecules. This leads to “proteome malfunction”—a state where proteins misfold and aggregate. These cellular errors are the suspected drivers of modern epidemics, including Alzheimer’s, Parkinson’s, and Type 2 Diabetes.
“Exposure to increasing atmospheric CO2 ‘is likely to lead to proteome malfunction’ – due to ‘protein misfolding, aggregation, charge distribution, and altered interaction with other molecules.'”
7. Conclusion: A New Urgency for Every Breath
The NHANES data suggests we have moved past the era of environmental theory and into a period of permanent physiological change. With essential minerals projected to hit their minimum healthy levels by 2099 and 2085, the internal stability of the human species is no longer a given. Our blood is documenting a crisis that our policy discussions have yet to fully grasp.
The fundamental question is whether human biology can adapt at the breakneck speed with which we are altering the air. Our systems were built for a 280 ppm world, and the current momentum of CO2 retention suggests we are entering uncharted territory. The alarm needs to be raised immediately; we are not just changing the climate of the planet, we are changing the chemistry of ourselves.
The infographic was generated by Notebook LM.
Larcombe, A.N., Bierwirth, P.N. Carbon dioxide overload, detected in human blood, suggests a potentially toxic atmosphere within 50 years. Air Qual Atmos Health 19, 44 (2026). https://doi.org/10.1007/s11869-026-01918-5
The Kids Research Institute Australia. (2026) Rising carbon dioxide levels now detected in human blood. Physics Today. https://phys.org/news/2026-02-carbon-dioxide-human-blood.html

Leave a comment